 
This animation below shows how to use the calculator to create a calculated column, but does not show what equation you will need to calculate the amount of magnesium consumed. Create a new calculated column to convert the volume of hydrogen gas produced into the amount of magnesium that has been consumed in the reaction. Remember, since this file is in the archive, you can no longer submit it.7. In order to receive a satisfactory score, you must answer all questions and provide explanations for your answers as requested. What other strategies do you use to learn chemistry? Some questions you might answer are: How are you using the lecture notes? Is this different from how you used them when preparing for Exam 1? How much homework have you done? Is this a higher or lower percentage than what you did from chapters 1-3? What about the practice exams? Textbook usage? Mentoring? Hint: its molar mass is not 16 g/mol.)Įxam Preparation Check-up-How's it going?Įxplain how your preparation for Exam 2 is going. Oxygen in the above equation is diatomic oxygen, O 2. How many grams of oxygen form when 5.3 grams of sodium chlorate decompose? (Be careful. How many moles of oxygen form when 2.2 moles of sodium chlorate decompose? How many moles of oxygen, O 2, form when 4 moles of sodium chlorate decompose? What conversion factor(s) did you use? Explain with words without using equations. How many moles of sodium chloride form when 4 moles of sodium chlorate decompose?Įxplain how you arrived at your answer. Sodium chlorate decomposes into sodium chloride and oxygen according to the following chemical equation: Rounding was done at the end to give us the appropriate number of significant figures in our answer. An extra significant figure was kept in each step to avoid rounding errors. We would report the answer as 16 g lithium. Next, you would need to convert moles of lithium nitride to moles lithium (using the mole ratio from the balanced chemical equation.)įinally, you would convert moles lithium to grams lithium (using the molar mass of lithium as a conversion factor). How many grams of lithium can be formed from 27 grams of lithium nitride according to the following chemical equation?įirst, you would need to convert the 27 g of lithium nitride to moles of lithium nitride (using the molar mass of lithium nitride as a conversion factor). How many moles of Fe 2O 3 are in 75 grams Fe 2O 3? What is the mass in grams of 4.5 moles of O 2? The molar mass of O 2 is 2 x 16.00 or 32.00 grams (1 mole O 2 = 32.00 grams O 2). 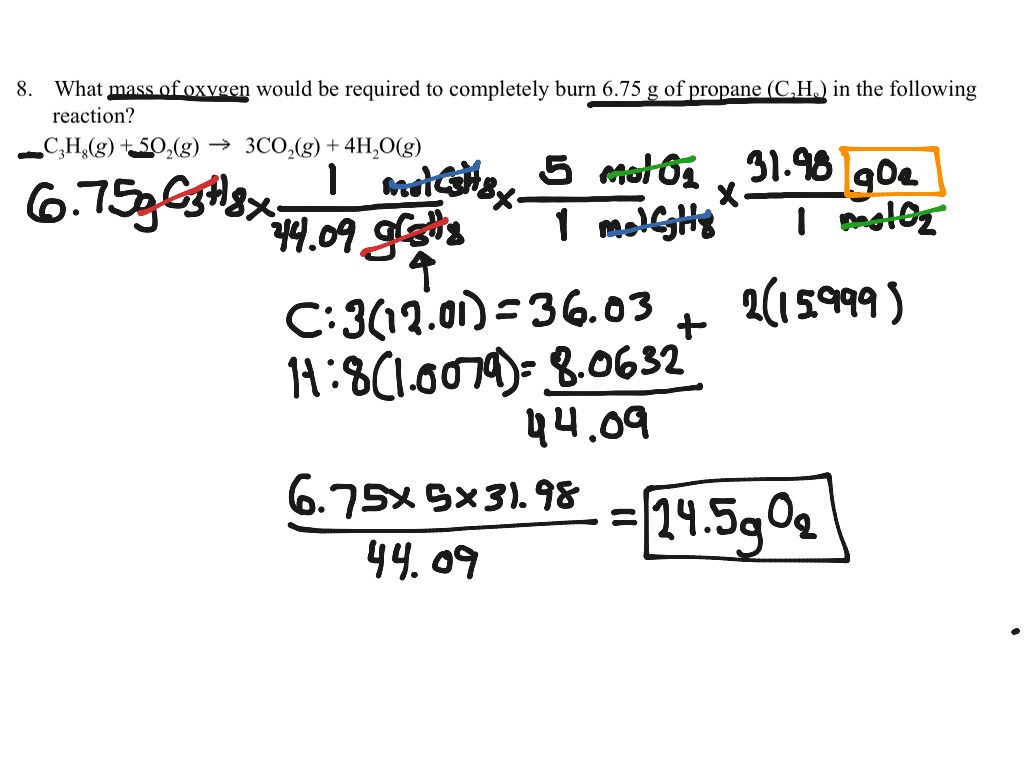
The molar mass of Fe is 55.845 grams (1 mole Fe = 55.845 grams Fe). Instead, we weigh the substance and calculate the corresponding number of moles using the molar mass. However, one can't just go to the shelf and count out numbers of atoms or numbers of moles of a substance. The ability to convert from moles of one substance to moles of another substance using mole ratios from balanced chemical equations is a useful skill. If you want to produce 6 moles of iron (III) oxide, how many moles of iron do you need to start with? (Assume you have all the oxygen that you need.) How moles of iron (III) oxide can be produced from 6 moles of iron? How would you set up the calculation? Which mole ratio would you use? If you want to know the number of moles of oxygen needed to completely react with 6 moles of iron, you would see from the balanced chemical equation that 3 moles of oxygen are needed for every 4 moles of iron. These mole ratio conversion factors can be used in many calculations. These mole ratios can be used as conversion factors. Or 4 moles of iron atoms react with 3 moles of oxygen molecules to yield 2 moles of iron (III) oxide. The balanced chemical equation for this process is:įrom the balanced chemical equation 4 iron atoms react with 3 oxygen molecules to yield 2 iron (III) oxide formula units. Rust, iron (III) oxide, forms when iron and oxygen react. 
The second objective is find out how your Exam 2 preparation is going.Ī balanced chemical equation indicates the relative numbers of molecules involved in a chemical reaction. The first objective is to solve problems using balanced chemical equations. 
The assignment this week has two objectives. When you are satisfied with your responses, click the SUBMIT button at the bottom of this page. You may change your mind as often as you wish. (in case your answer gets used in class). Please type the last five digits of your ID number: This assignment was due by noon on Tuesday, March 11, 2003. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
